 
The bond can be considered covalent, composed of a pair of shared electrons. Picture a bond between a sodium atom and a chlorine atom. Each of these elements has a very low electronegativity when compared with fluorine, and the electronegativities decrease from lithium to cesium. It is usually measured on the Pauling scale, on which the most electronegative element (fluorine) is given an electronegativity of 4.0 ( Table A2).Ī graph showing the electronegativities of the Group 1 elements is shown above. However, the distance between the nucleus and the outer electrons increases down the group electrons become easier to remove, and the ionization energy falls.Įlectronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. As mentioned before, in each of the elements Group 1, the outermost electrons experience a net charge of +1 from the center. the distance between the outer electrons and the nucleus.ĭown the group, the increase in nuclear charge is exactly offset by the increase in the number of inner electrons.the amount of screening by the inner electrons,.Ionization energy is governed by three factors: Notice that first ionization energy decreases down the group. \Ī graph showing the first ionization energies of the Group 1 atoms is shown above. The first ionization energy of an atom is defined as the energy required to remove the most loosely held electron from each of one mole of gaseous atoms, producing one mole of singly charged gaseous ions in other words, it is the energy required for 1 mole of this process: Therefore, the atoms increase in size down the group. More layers of electrons take up more space, due to electron-electron repulsion. The only factor affecting the size of the atom is the number of layers of inner electrons which surround the atom. This is true for each of the other atoms in Group 1. This effect is illustrated in the figure below: The positive charge on the nucleus is canceled out by the negative charges of the inner electrons. In each element, the outer electron experiences a net charge of +1 from the nucleus. The attraction the outer electrons feel from the nucleusĬompare the electronic configurations of lithium and sodium:.The number of layers of electrons around the nucleus. 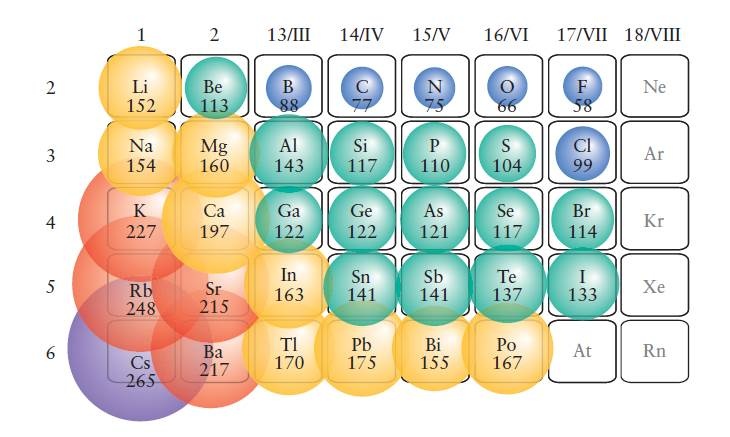
The radius of an atom is governed by two factors: The chart below shows the increase in atomic radius down the group. It is very radioactive and used as an industrial gamma ray source.\) Another isotope, 137Cs is not made naturally but is made after nuclear fission has been done. 133Cs is used in atomic clocks, its vibration frequency used to define the length of the second. Therefore, the naturally-occurring isotope of caesium is 133Cs, which is not radioactive. Isotopes and compoundsĬaesium has at least 39 known isotopes ranging in atomic mass from 112 to 151.  
They named this substance caesium, after the color blue. Because of these lines, they concluded that in addition to the elements already found, there must be another unknown substance in the mineral water. After they separated calcium, strontium, magnesium and lithium, they saw two lines in the "blue" range of the spectrum. They were testing mineral water, from Bad Dürkheim. Caesium was first described in 1861, by Gustav Robert Kirchhoff and Robert Wilhelm Bunsen. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
